Services: Enzymatic DNA for AAV Production
neDNA™ can address challenges in pDNA manufacturing for AAV production. neDNA™: enzymatic DNA for AAV Production
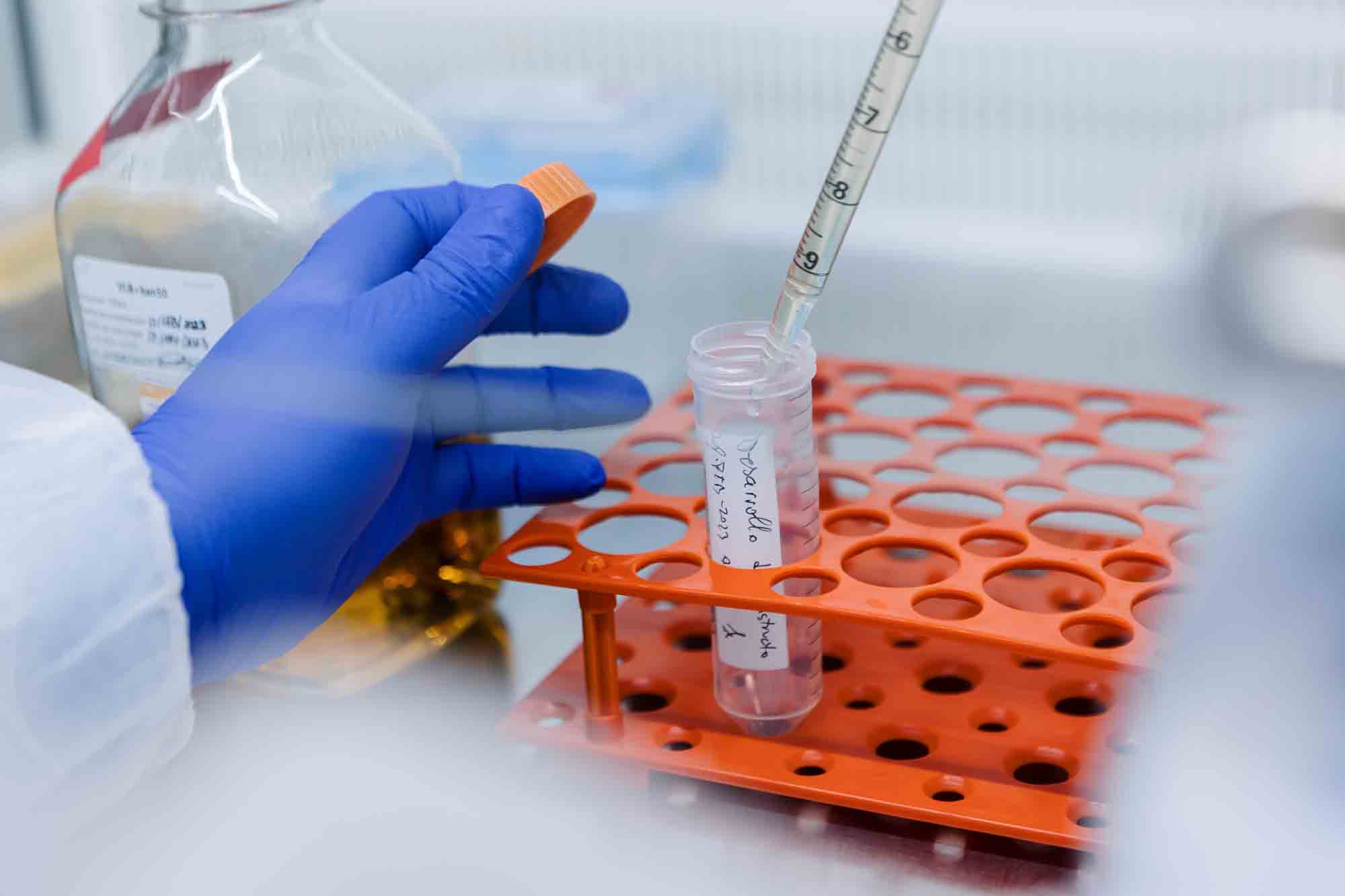
At TAAV we produce neDNA™: the first option for critical starting material DNA for AAV production. Designed to transform and accelerate the traditional gene therapy development timelines, this enzymatic DNA offers an alternative to plasmids for rAAV manufacturing.
RAPID PRODUCTION
Enzymatic process.
Industrial scalability (smaller footprint).
Shorter production timelines.
Gram-scale batches in 4 days. (GMP-compliant)
RELIABLE COMPLEX SEQUENCES AMPLIFICATION
Phi29 amplifies complete sequences, including ITRs, GC rich regions, polyA.
Linear and reproducible yield, batch homogeneity.
INTENTED TO IMPROVE SAFETY PROFILE
No bacterial sequences (no antibiotic resistance gene & no replication origin).
Suggestive of a reduced risk of immunogenicity.
Grades: Enzymatic DNA Production for AAV
Phased-appropriate grade material, working at all stages of clinical development and quantities needed.
TAAV manufactures three different grades of neDNA™ in our facilities: Research, Pre-Clinical and Clinical Grade. We are specialized in the production of Enzymatic DNA for AAV production
Three types of neDNA™ are currently manufactured at TAAV: RepCap (C), Helper (H), and transgenes (T).
neDNA™: Enzymatic DNA production for AAV.


ORIGEN GRADE
Intended for easy project kick-off.
neDNA™ delivery: 3-5 mg.
Support candicate selection.
Analytical Report & Project Report provided with delivery.
R&D GRADE
Suggested for evaluation/feasibility studies.
Scales available from 5-20mg.
Produced with representative manufacturing process of higher scales.
Analytical Report provided with delivery.
PRECLINICAL GRADE (research)
Intended for process development, research and toxicology studies.
Scales available starting at 50mg.
Produced with identical process in the same facility as GMP-compliant grade.
Full batch record documentation and QA oversight with set of specifications.
GMP-COMPLIANT GRADE
Intended for toxicology studies, clinical and commercial production.
Scales available from 100mg to multiple grams.
Produced under ISO classified production suites following GMP principles.
Full batch record documentation and QC/QA release with set of specifications.
How we work
TAAV is CDMO working with biopharma companies, research centers and entities developing programs based on rAAV-based gene therapies. We offer neDNA™: Enzymatic DNA Manufacturing for rAAV production.
We are a trusted partner for those who are committed to advancing the field of gene therapy. We all are leading by our passion to collaborate in the development and manufacture of new gene therapies that improve the quality of life of society and especially of those who suffer from pathologies and rare diseases.
At TAAV we aim to understand the unique requirements of each client. We are a science and business minded company. Partner with us and let’s make it real together.
We have a common goal: to improve the patients' quality of life and make the world a better place.
Contact our team to learn more about how neDNA™ can be the first option for critical starting material for rAAV manufacturing.


